Dr. Vikas Singh is a forward-thinking Consultant Urologist in Indore, at the forefront of innovative treatments for Urethral Stricture Disease. He is among the select few specialists in Central India offering AALBEC (Autologous Adipose and Buccal Epithelial Cell) therapy—a revolutionary regenerative medicine approach combining stem cell technology with traditional reconstructive principles. This cutting-edge technique uses the patient’s own cells (harvested from buccal mucosa and adipose tissue) to create a bioengineered graft that promotes natural urethral healing and regeneration with minimal invasiveness. Practicing at Kokilaben Dhirubhai Ambani Hospital Indore with access to specialized tissue engineering facilities and multidisciplinary regenerative medicine support, Dr. Singh offers patients an innovative alternative that bridges the gap between temporary endoscopic procedures and complex open reconstruction. This minimally invasive approach shows promising results for select stricture cases while preserving future treatment options. We maintain complete transparency regarding this emerging technology, treatment costs, and provide comprehensive guidance for our patients across Central India.
Consultant Surgeon at Kokilaben Dhirubhai Ambani Hospital, Indore, with advanced regenerative medicine capabilities.
Among the first urologists in Central India to offer cell-based regenerative therapies for stricture disease.
Expertise across all treatment modalities from endoscopic to regenerative to open reconstruction.
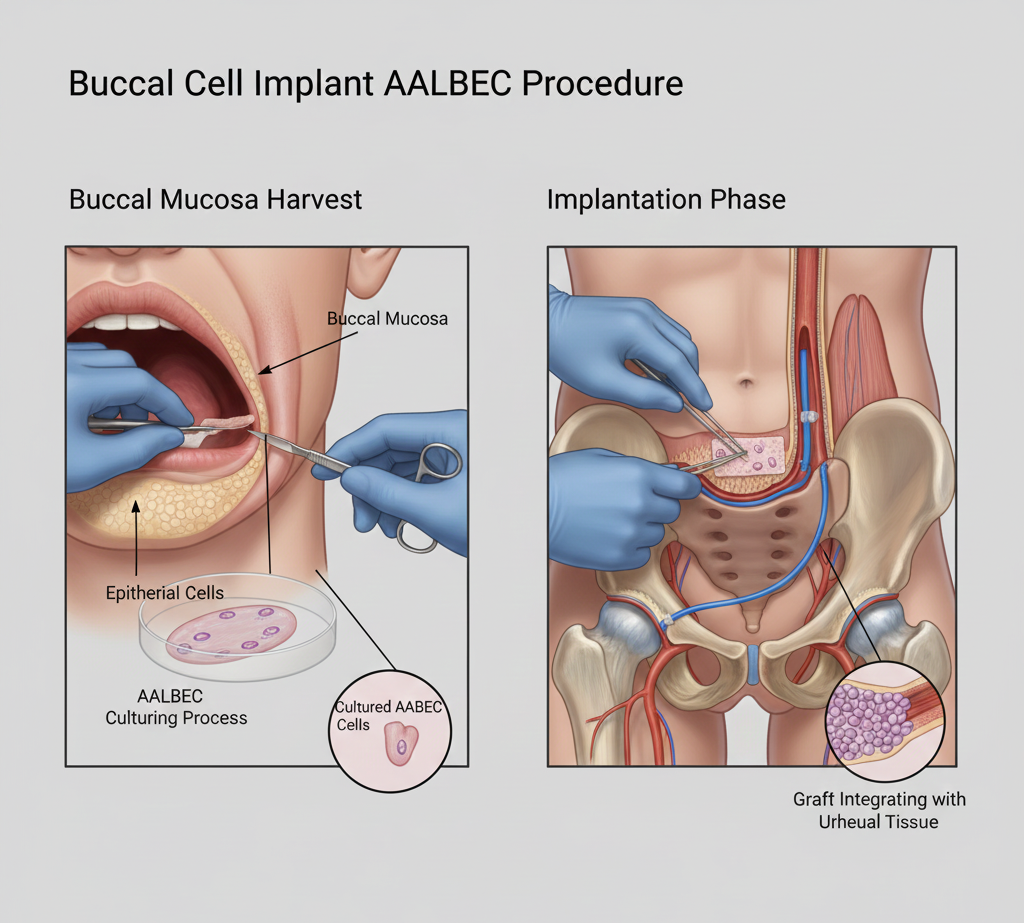
AALBEC represents a paradigm shift in stricture treatment, moving from mechanical solutions (cutting scar tissue or replacing it with grafts) to biological regeneration. The therapy uses autologous (your own) cells—buccal epithelial cells from the inside of your cheek combined with adipose-derived stem cells from fatty tissue—to create a living, bioengineered construct that integrates into the urethral tissue and promotes natural healing with minimal scar formation.
Adipose-derived stem cells possess remarkable regenerative properties: they secrete growth factors that promote tissue healing, reduce inflammation and scar formation, enhance blood vessel development (angiogenesis), and differentiate into various cell types needed for tissue repair. Combined with buccal epithelial cells (which naturally line moist surfaces like the urethra), this creates an optimal biological environment for urethral regeneration rather than scar tissue formation.
AALBEC is particularly promising for patients with recurrent short to moderate strictures (<3-4 cm) who have failed endoscopic treatments but want to avoid major open reconstruction, patients with strictures in challenging locations, those seeking minimally invasive options with faster recovery, and patients with medical conditions making extensive surgery risky. It's also valuable for preserving future options—if AALBEC doesn't achieve desired results, traditional urethroplasty remains fully available.
The procedure begins with harvesting your own cells under local anesthesia. A small sample of buccal mucosa (3-4 cm²) is taken from inside your cheek, and a small amount of adipose tissue (fat) is obtained through a minor liposuction technique, typically from the abdomen or thigh. These tissues contain the epithelial cells and stem cells needed for the therapy. The donor sites heal quickly with minimal discomfort.
The harvested tissues are processed in a specialized laboratory where buccal epithelial cells are cultured and expanded, and adipose-derived stem cells are isolated and activated. These cells are then combined with a biocompatible scaffold material to create a living graft construct. This processing typically takes 2-4 weeks, during which the cells multiply and the engineered tissue matures.
Once the bio-engineered graft is ready, it’s implanted using a minimally invasive endoscopic approach. Under anesthesia, the strictured segment is accessed through the urethra using specialized instruments. The AALBEC construct is precisely positioned at the stricture site where it integrates with the surrounding tissue. A catheter remains in place for 2-3 weeks to support healing. The entire implantation procedure takes 1-2 hours with minimal trauma to surrounding structures.
This innovative approach offers biological healing rather than mechanical intervention. It's minimally invasive (endoscopic implantation), uses your own cells (no rejection risk), promotes natural tissue regeneration with less scarring, provides faster recovery than open surgery (1-2 weeks vs. 6-8 weeks), and preserves all future treatment options if additional intervention is needed. Success rates are still being established but show promise in 60-75% of appropriate candidates. Primary Advantages: Biological regeneration, minimal invasiveness, preserves future options, faster recovery than open surgery.
Simple mechanical incision of scar tissue with high recurrence rates (40-60%). Quick recovery, but doesn't address underlying pathology—scar tissue often reforms. Multiple procedures are typically needed. Key Limitation: Doesn't prevent re-scarring; a temporary solution for many patients.
Gold standard with highest success rates (85-95%) but requires open surgery, longer recovery (6-8 weeks), and more invasive tissue manipulation. Excellent for complex cases but may be more intervention than some patients need or want initially. Key Limitation: Most invasive option; best reserved for cases where less invasive approaches have failed or aren't suitable.
Promotes natural tissue healing rather than leaving scar tissue or foreign material—the urethra heals with your own regenerated tissue.
Endoscopic approach avoids external incisions, reduces surgical trauma, and minimizes risk to surrounding structures.
Return to normal activities within 2-3 weeks—significantly faster than open reconstruction while potentially more durable than simple OIU.
If AALBEC doesn't achieve desired results, traditional urethroplasty can still be performed without any disadvantage—nothing is "burned" or damaged.
Schedule a private, confidential consultation with Dr. Singh in Indore.
AALBEC represents a middle ground between endoscopic procedures and open reconstruction in terms of both invasiveness and effectiveness. Early clinical data suggests success rates of 60-75% for appropriately selected patients—better than repeated OIU (which has 30-40% success after first failure) but not quite as high as open urethroplasty (85-95%). However, AALBEC offers advantages neither traditional approach provides: biological regeneration using your own cells, minimal surgical trauma compared to open surgery, and preservation of all future treatment options. For patients who’ve failed 1-2 endoscopic procedures but want to avoid major reconstruction, or for those with medical conditions making extensive surgery risky, AALBEC provides an excellent intermediate option. It’s important to understand this is still an evolving technology—Dr. Singh will provide realistic expectations based on current evidence and your specific stricture characteristics.
AALBEC is considered very safe because it uses autologous cells (your own tissue), which eliminates risks of rejection, allergic reactions, or disease transmission associated with donor tissue or synthetic materials. The cell harvesting procedures (buccal biopsy and small liposuction) are minor with minimal risks—primarily minor bleeding, temporary discomfort, or infection (rare). The laboratory processing follows strict sterility protocols to prevent contamination. Implantation risks are similar to other endoscopic urethral procedures: temporary urinary symptoms, potential infection, or rare perforation. Because the therapy promotes natural healing rather than leaving foreign material, long-term complications are minimized. The main “risk” is treatment failure—the stricture not improving adequately—in which case you haven’t lost anything and can proceed to traditional treatments. Importantly, AALBEC doesn’t make future urethroplasty more difficult if needed, unlike repeated endoscopic procedures which worsen scarring.
The complete AALBEC journey spans approximately 6-8 weeks from initial consultation to full recovery. Week 0-1 involves comprehensive evaluation and the cell harvesting procedure (outpatient, 30-45 minutes). Weeks 1-4 comprise laboratory processing where your cells are cultured, expanded, and bio-engineered into the therapeutic construct—you continue normal activities during this period. Week 4-5 is the implantation procedure (1-2 hours, minimal hospital stay) with catheter placement. Weeks 5-7 involve catheter removal and initial recovery. By weeks 6-8, most patients have returned to full normal activities. However, complete tissue integration and remodeling continues for 3-6 months, with ongoing monitoring to assess long-term success. While this timeline is longer than simple OIU (which takes just days), the potential for more durable results with minimal invasiveness makes it worthwhile for appropriate candidates.
Insurance coverage for AALBEC is variable and evolving as this is an emerging regenerative medicine technology. Currently, coverage depends on your specific insurance policy, the indication for treatment (medical necessity), and how the procedure is coded. Some insurance companies cover it under experimental or innovative treatment provisions with prior authorization, while others may not yet have specific coverage policies. Government schemes like Ayushman Bharat may have limitations on coverage for newer technologies. Our administrative team has experience navigating insurance approval for advanced treatments and will work with your insurance provider to maximize coverage possibilities. We also provide transparent cost estimates upfront (typically higher than simple OIU but less than complex open urethroplasty) and offer guidance on payment options if insurance coverage is partial or unavailable. The investment often reflects the sophisticated laboratory processing and bio-engineering required for cell-based therapy.
AALBEC’s primary advantage over synthetic materials or cadaveric grafts is that it uses your own living cells, which integrate naturally and continue to function as part of your body. Synthetic scaffolds (like various biomaterials being researched) provide structural support but don’t have living cells to promote active healing—they rely on your body’s cells to eventually populate the scaffold, which may not happen optimally. Cadaveric grafts (from deceased donors) can work but carry theoretical risks of rejection or disease transmission and don’t have the regenerative stem cell component. Traditional buccal mucosal grafts (used in standard urethroplasty) work well but require more invasive harvesting and implantation through open surgery. AALBEC combines the biological advantages of your own cells, the regenerative power of stem cells, and the minimally invasive delivery method—offering a unique combination not available with other materials. The stem cell component particularly sets AALBEC apart by actively promoting healing rather than serving as passive material.
One of AALBEC’s significant advantages is that treatment failure doesn’t compromise future options. If the therapy doesn’t achieve adequate improvement—assessed through follow-up uroflowmetry and symptom evaluation at 3-6 months—you can proceed to traditional treatments without any disadvantage. The minimally invasive nature means no additional scar tissue has been created, no tissue has been excised, and the stricture anatomy hasn’t been made more complex. You could then undergo standard urethroplasty with buccal mucosal graft or other appropriate technique with the same success rates as if AALBEC hadn’t been attempted. This “preserving future options” characteristic makes AALBEC an attractive option for patients wanting to try a less invasive approach before committing to major reconstruction. Dr. Singh will work with you to determine appropriate timing for assessing results and making decisions about additional treatment if needed.
The leading center for advanced, compassionate urological care and treatment in Indore, Madhya Pradesh.